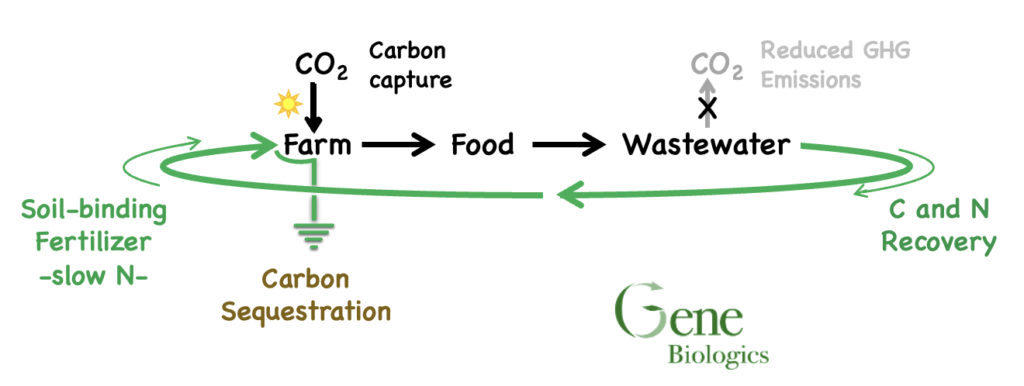
We are developing a renewable and regenerative way to fertilize plants where the fertilizer remains tightly bound to soils, decreases N pollution, creates a carbon sink and lowers greenhouse gas emissions.
How this works: Plants fix CO2 from the air. Some of this captured carbon is currently lost as greenhouse gases (GHG) during wastewater breakdown. We redirect this carbon back to soil/plants by converting wastewater to a slow-release protein fertilizer. This protein has a polyarginine component that binds very tightly to the mineral portion of soil.
The net effect is to continuously pull carbon from the atmosphere and bind it to soil
Farmers create long-term carbon management by fertilizing plants with our protein. When applied, the water-soluble protein migrates into the root zone and binds particles of clay or is slowly degraded and taken up by plants and bacteria. Any unused protein is retained in the soil. As more protein is added, excess protein binds deeper mineral layers creating a nutrient-rich reservoir. Based on initial tests with our polyarginine fertilizer, 5-10 gigatons of additional carbon could be sequestered in just the top one meter of Earth’s soil. Humans currently produce about 30 gigatons too much CO2 per year, so our product could make a sizeable dent in avoiding climate change.
Expedited, self-sequestering soil carbon. All without costs for capture or for injection of CO2 underground; carbon sequestration on a global scale while rejuvenating and making healthier soils
Enhanced carbon sequestration in soils
 Normal carbon residue, such as from corn stalks left fallow, is termed particulate carbon and is vulnerable to erosion and breakdown. Typically 90% of particulate carbon is lost over time.
Normal carbon residue, such as from corn stalks left fallow, is termed particulate carbon and is vulnerable to erosion and breakdown. Typically 90% of particulate carbon is lost over time.
The soil-binding part of our peptide consists of multiple arginine amino acids which have one of Nature’s most strongly positive organic charges. Because soil is negatively charged, our water-soluble peptide travels into and tightly binds the stable, mineral portion of soil -exactly where organic carbon is seen in old soils. In very old soil samples (10,000 years and more) the remaining soil N appears to be largely peptidyl in origin (1) with about 1/3rd of the carbon likely coming from protein breakdown.
We’ve shown that our peptide travels into and binds the clay in soils so tightly that it remains bound even after washing with 1,000 equivalent volumes of water.
This expedited delivery of carbon happens without changing how farms are managed and this C is in addition to any carbon already being sequestered by crops. This will work with all crops and soil types; forests, no-till, and commercial crops alike.
Changing the way society uses vs loses nutrients
In addition to C sequestration, our process lowers societal GHG emissions in ten ways: less CO2 from less industrial Ag NH3 production; less N2O loss from soils; less soil CO2 loss from no-till ag; less CO2 due to single application of fertilizer; less CO2 from more efficient WWTP electricity use; capture of biogenic carbon from wastewater treatment; less CH4 and N2O from wastewater treatment; and, less CH4 and N2O from landfills.
Compare our product to the current industrial approach to N fertilizer manufacture, use and discharge. Currently, methane (CH4) in natural gas is converted to NH3 fertilizer, food and pollution. The overall process emits ~400 million metric tons of CO2 and CO2 equivalents (CO2 e) per year for just the US. Most of the CO2e is actually due to excess N waste:
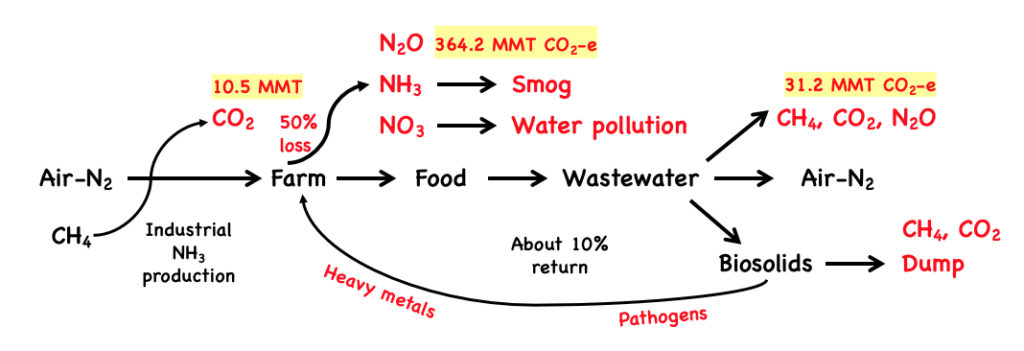 The cost of US agricultural nitrogen pollution to both human health and the environment is about equal to the GDP from America’s farms; some $150 billion/year (2). This estimate doesn’t include the costs of CO2, NH3, N2O and CH4 as direct/indirect greenhouse gases. These GHG losses are shown in red in the above figure and are derived from the EPA draft report for CO2 inventories.(6) These emissions are driven by the ~50% loss of nitrogen (3,4) from synthetic fertilizer use and represent large direct costs to the farmer. For example, an Iowa corn farmer on poor Iowa soil spends about $70/acre on N fertilization, and takes in $70/acre in net profit.(7) Avoiding this N loss could increase this farmer’s income by some 30%. Farmers will save money while saving our air, water, and soils.
The cost of US agricultural nitrogen pollution to both human health and the environment is about equal to the GDP from America’s farms; some $150 billion/year (2). This estimate doesn’t include the costs of CO2, NH3, N2O and CH4 as direct/indirect greenhouse gases. These GHG losses are shown in red in the above figure and are derived from the EPA draft report for CO2 inventories.(6) These emissions are driven by the ~50% loss of nitrogen (3,4) from synthetic fertilizer use and represent large direct costs to the farmer. For example, an Iowa corn farmer on poor Iowa soil spends about $70/acre on N fertilization, and takes in $70/acre in net profit.(7) Avoiding this N loss could increase this farmer’s income by some 30%. Farmers will save money while saving our air, water, and soils.
Sustainable N and C recovery from waste
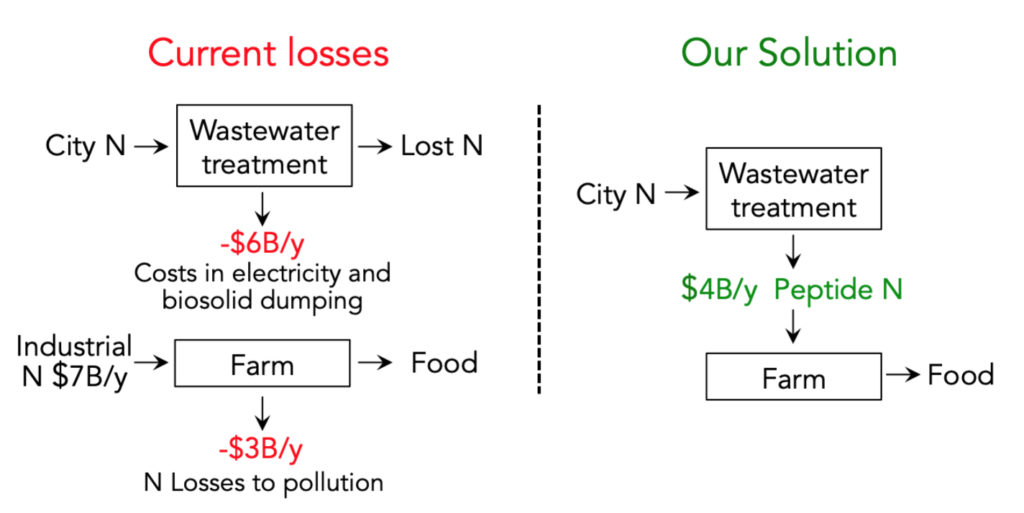
Wastewater arriving at a city municipal wastewater treatment plant (a Water Resource Recovery Facility or WRRF) contains a lot of beautiful nitrogen (from a growing plant’s point of view), much of it in the form of ammonium, NH4+. However, it can’t just be shipped back to farms because it is dilute, costly to ship, and may contain pathogens and other toxins requiring removal or treatment. Instead, in the US we spend $6B/y in converting the valuable N input in wastewater into worthless N2 gas, greenhouse gases, and costly-to-dispose bacterial cell mass (biosolids). In fact, treatment of N in wastewater (about $4800/metric ton) (5) costs ~10x more than the intrinsic value of the N as urea.
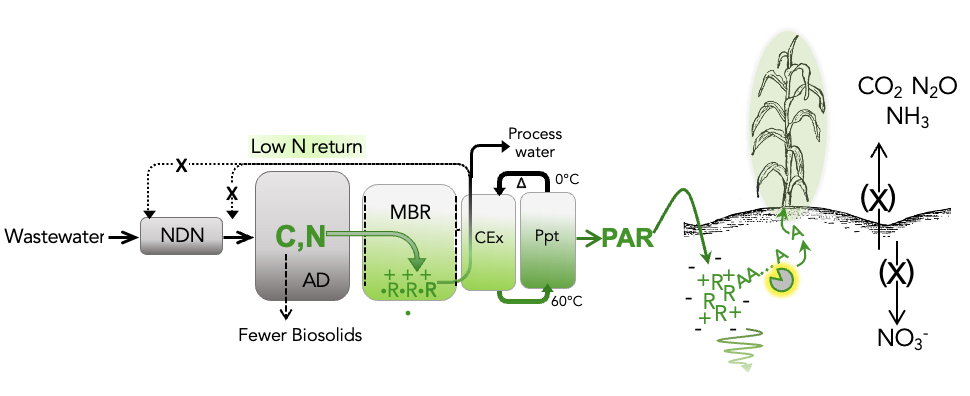 Our patented process is called Nitrogen Arginine Biorecovery (NAB). It fits onto the back end of typical WRRFs, and recovers the N as a sterile, pure product that binds soil for slow release to plants. The process leverages the same strong positive charge used for binding soil in order to cheaply pull polyarginine-rich proteins (PAR) out of waste streams and purify it. This is noteworthy, since pulling anything out of wastewater besides methane (low value) or biosolids (negative value) is a significant accomplishment.
Our patented process is called Nitrogen Arginine Biorecovery (NAB). It fits onto the back end of typical WRRFs, and recovers the N as a sterile, pure product that binds soil for slow release to plants. The process leverages the same strong positive charge used for binding soil in order to cheaply pull polyarginine-rich proteins (PAR) out of waste streams and purify it. This is noteworthy, since pulling anything out of wastewater besides methane (low value) or biosolids (negative value) is a significant accomplishment.
Our process continuously removes wastewater N and C as protein. This limits NH4+ inhibition of the WRRF anaerobic digestion treatment step, resulting in less biosolid disposal and decreased NDN (nitrification/denitrification) treatment. NAB relies entirely on environmentally friendly solutions that are suitable for reuse and compatible with wastewater treatment. Overall, we anticipate a ~10% reduction in WRRF capital and operational expenses (this includes PAR production costs). Per design, the final product is a sterile and odor-free powder ready for shipment back to farms. One truckload of product saves the equivalent of 23 truckloads of de-watered biosolids that cities would otherwise have to landfill or disposed of.
There is no “green premium”. We expect our peptide to be priced competitive with slow-release urea formulations. We can do this because the product more than pays for itself through WRRF process savings, largely by pulling N out from solution as a solid, increasing anaerobic digestion throughput and decreasing biosolid transport costs.
The US currently spends and loses about $16B/y on N fertilizer. Full application of NAB to WRRF could account for one quarter of all current industrial N fertilizer production; a ~$4B market in the US.
- Arginine has a very strong positive charge (pKa >12.5) while soil is negatively charged. Our polyarginine peptide has a string of arginines, each of which binds collectively to neighboring negative charges
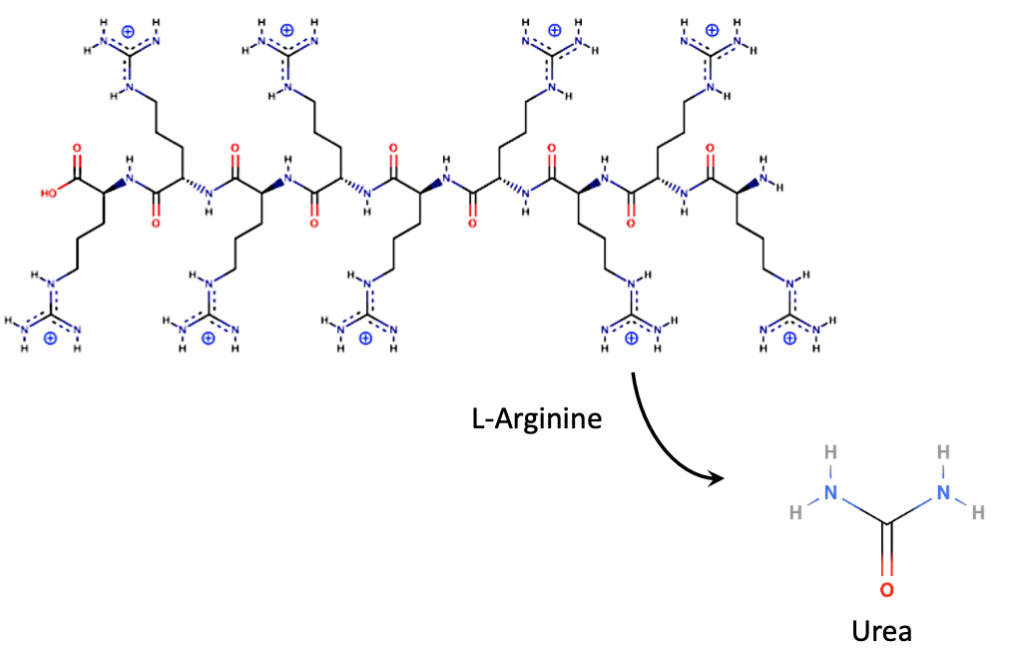 in the surrounding soil. The peptide sticks to the soil and becomes less available to enzymatic break-down for stability in soil. Non-arginine peptide sequence (the part of our peptide containing amino acids other than arginine) should break down faster for timed N release (see the yellow “pacman” enzyme shown in the NAB diagram above). Because of the high pKa this should work on any clay-containing farm soil regardless of pH.
in the surrounding soil. The peptide sticks to the soil and becomes less available to enzymatic break-down for stability in soil. Non-arginine peptide sequence (the part of our peptide containing amino acids other than arginine) should break down faster for timed N release (see the yellow “pacman” enzyme shown in the NAB diagram above). Because of the high pKa this should work on any clay-containing farm soil regardless of pH. - We directly monitor the polyarginine C-sequestering portion of the protein in soil by a one-step stripping and fluorescence-based assay for immediate carbon verification.
- Our peptide acts as a unique slow-release “carrier” of urea When the polyarginine peptide does break down, the individual amino acids can either be taken up directly by plants, or can be converted by soil organisms to the amino acid ornithine and urea.
- Current “green” industrial slow-release N fertilizers plasticize the urea or add an agent to kill or inhibit soil organisms in order to create slow-release nitrogen fertilizer. We use the inherent charge of soil to attain slow release of N. No expensive coatings, no inhibitors.
- Our peptide can be applied to soil either as a solid or liquid, since the peptide is ~50% soluble in water by weight.
- The peptide does acts as a fertilizer, in particular when delivered hydroponically. It is less toxic to plants at high application rates than urea or NH4Cl, so it may need only one application (for instance when seeding).
- The peptide should be ideal for no-till fertilization. In this way, farmers create better soils in part by not having to till, and in part by securing N in a non-volatile and non-leaching manner.
- Polyarginine is a storable, marketable solid. Typically, large farm animal operations produce way more nitrogen from manure than nearby fields can utilize. The nitrogen is literally and purposefully wasted; it is allowed to sit in lagoons until excess N is vaporized off as NH3. There are odor and safety issues relating to NH3 release. With our process, excess N is instead converted to off-gate farm revenue for direct application to fields/crops when needed. This makes for less agricultural N pollution and more efficient farming.
NAB has the potential to generate verifiable carbon credits and make a significant positive impact. The components of this process are currently undergoing refinement through collaboration with a national laboratory. Please check back and, if you have questions, contact Matt Wecker!
- (1) GB Curry, et al., Amino acid distribution in a loess-palaeosol sequence near Luochuan, Loess Plateau, China. Organic geochemistry, 1994. https://www.sciencedirect.com/science/article/abs/pii/0146638094901759
- (2) Sobota, D.J., et al., Cost of reactive nitrogen release from human activities to the environment in the United States. Environmental Research Letters, 2015. 10(2): p. 025006.
- (3) Meisinger, J. and G. Randall, Estimating nitrogen budgets for soil-crop systems. Managing nitrogen for groundwater quality and farm profitability, 1991(managing nitrogen): p. 85-124.
- (4) Hermanson, R., et al., Nitrogen use by crops and the fate of nitrogen in the soil and vadose zone. Washington State University and Washington Department of Ecology Interagency Agreement, 2000(C9600177).
- (5) Municipal Nitrogen Removal Strategies. Table 3-5. Average of 8 WTTPs. https://www.epa.gov/sites/default/files/2019-08/documents/municipal_nutrient_removal_technologies_vol_i.pdf
- (6) Draft Inventory of U.S. Greenhouse Gas Emissions and Sinks: 1990-2019
- (7) 2018 Purdue Crop Cost & Return Guide